
YOUR FITNESS BLOG
Weight Gain Results From Lack of Sleep
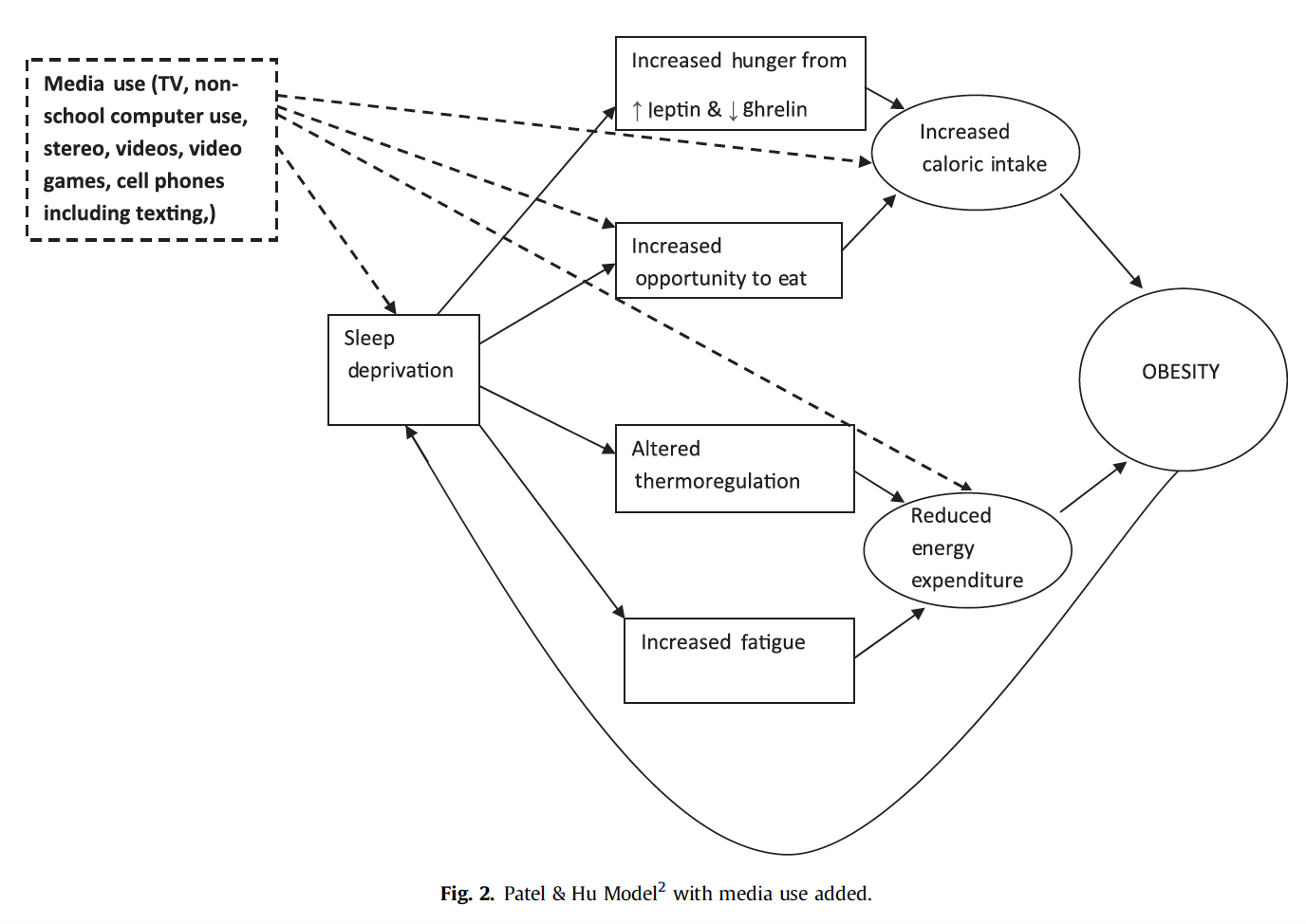
In a previous article, we identified that changes in body weight occur with alterations in energy balance. For example, when energy intake exceeds energy expenditure, net energy balance is positive and weight gain ensues. While the majority of public health intervention and research focuses on inactivity and unhealthy diet as contributors to weight gain, multiple studies show a correlation between insufficient sleep and weight gain in adults and children. The potential mechanisms related to sleep deprivation and positive energy balance include:
- Altered thermogenesis
- Increased fatigue contributing to physical inactivity
- Alterations in appetite with increased opportunity for eating
- Alterations in appetite regulating hormones
Altered Thermogenesis
Multiple studies demonstrate that sleep deprivation alters thermoregulation by increasing 24-hour total daily energy expenditure (TDEE) [3, 4]. A study by Markwald and colleagues (2013) sleep deprived adult women to 5-hours of sleep for 5 days to examine the effects of insufficient sleep on energy expenditure and food intake [3]. This study showed a ~5% increase (~111 kcal/d) in 24-hour TDEE on the recovery day following sleep deprivation [3]. In support, Jung and colleagues (2011) found a ~7% increase in 24-hour TDEE during the recovery day following sleep deprivation [4].
Despite increases in energy expenditure during wakefulness, increases in food intake and decreased physical activity from insufficient sleep creates a positive net effect that promotes weight gain [3, 12]. Jung and colleagues showed that a ~7% increase in TDEE during wakefulness is nearly offset by a ~5% decrease in TDEE the following day, resulting in a 2% net cost [4]. Increasing food intake would create an even larger positive energy balance that would completely offset any increases in energy expenditure [12].
Increased Fatigue Contributing to Physical Inactivity
It is well established that chronic sleep deprivation leads to increased feelings of fatigue. Fatigue can encompass sleepiness, referred to as the tendency to fall asleep, and further expand to include physical and mental fatigue or tiredness [1]. Subjective reports of increased sleepiness are documented after only one night of sleep deprivation with cumulative sleep deprivation leading to even greater levels of tiredness [14]. Also, deficits in cognitive performance have been found in healthy adults sleep restricted to 6-hours of sleep for 14 consecutive nights [14].
Subjects that report increased daytime sleepiness show a significant reduction in physical activity [2, 15]. Schmid and colleagues (2009), showed that healthy, adult men sleep deprived for 4 hours of sleep over 2 nights significantly decreased physical activity following each night of sleep curtailment [8]. One study showed a 5% reduction in energy expenditure during the recovery day following sleep deprivation [4]. Also, following poor sleep, there is a shift from higher physical activities towards that of lower intensity activities, which may, over a longer period, promote weight gain from decreased energy expenditure [8].
Alterations in Appetite with Increased Opportunity for Eating
One pathway by which sleep deprivation may contribute to weight gain is through increased caloric intake [7]. Compared to 10-hours of sleep, 4-hours of sleep per night over just two days showed a 24% increase in hunger and 23% increase in appetite, measured by validated visual analog scales [10]. Markwald and colleagues (2013) suggest that increased food intake during sleep deprived hours is a physiological adaptation to supply energy to sustain wakefulness [3]. However, food intake exceeds that of energy requirements resulting in weight gain [3].
Also, an increase in appetite is shown for more calorie-dense foods with high carbohydrate content such as sweet foods, salty and starchy foods [7]. This is compared against less hunger for fruits, vegetables and higher protein foods during sleep deprivation [7]. Increased appetite for high fat and high carbohydrate foods are associated with increases in appetite stimulate hormone ghrelin and a decrease in appetite suppressing hormone leptin [7].
Moreover, it is proposed that increase calorie consumption may result from increased opportunity to eat from being awake during sleep hours [9]. As calorie intake is likely proportional to the number of waking hours, additional wake-time with sleep deprivation may create an environment conducive to eating [9]. Increased opportunity to eat may be enhanced even more where wake-time is spent performing sedentary activities such as sitting and watching television, which promote snacking [9]. Sedentary activity combined with increased food intake during sleep hours would likely promote weight gain.
Alterations in Appetite Regulating Hormones
Two key appetite regulating hormones, leptin and ghrelin, play an important role in the interaction between sleep deprivation and weight gain [13]. As previously discussed in a separate blog post, leptin is a hormone produced by fat cells that provides important information about energy status to parts of the brain responsible for appetite suppression [11]. In contrast, ghrelin is a hormone produced in the stomach and other tissues that stimulate growth hormone secretion, gastric motility and communicates with the brain to stimulate appetite and food intake [5].
Short sleep duration of less than 8-hours has been associated with decreased leptin and increased ghrelin [11, 13]. Spiegel and colleagues (2004) showed that just 6 days of 4-hours of sleep in healthy, young subjects resulted in a 26% decrease in leptin levels [11].
Taheri and colleagues (2004) demonstrated similar findings in their longitudinal study of sleep disorders. In their study, subjects that habitually slept for 5-hours each night was associated with 15.5% lower leptin levels and 14.9% higher ghrelin levels compared to those sleep 8-hours on average each night [13]. These changes in leptin and ghrelin are likely to increase appetite and create a positive energy balance resulting in weight gain.
As a mechanism underlying alterations in leptin and ghrelin, it is suggested that sleep deprivation brings about increased sympathetic nervous system output leading to leptin reduction [13]. As the sympathetic nervous system is responsible for stress related responses, it may be that lack of sleep is perceived by the body as a stressor [13]. Cortisol, a hormone released in response to stress, is found to be higher following a night of reduced sleep compared with extended sleep durations, lending support to a potential biological stress response [13].
Summary
Sleep deprivation results in weight gain because energy intake from food exceeds that of energy expended during wakefulness. Weight gain is further exacerbated because sleep exhaustion results is reduced physical activity the following day. Finally, lack of sleep leads to reciprocal changes in appetite regulating hormones leptin and ghrelin that encourage consumption of calorie-dense foods. As a weight management strategy, it is suggested that for healthy adults, 7 to 9 hours of sleep per night is optimal [16].
For more information on our personal training services please click here to read more.
References:
[1] Akerstedt, T. Sleep Loss and Fatigue in Shift Work and Shift Work Disorder. Sleep Medicine Clinic. June. Vol. 4, No. 2, pp. 257-271.
[2] Briones, B. et al. 1996. Relationship Between Sleepiness and General Health Status. Sleep. Vol. 19, No. 7, pp. 583–588.
[3] Markwald, R. R. et al. 2013. Impact of insufficient sleep on total daily energy expenditure, food intake, and weight gain. Proceedings of the National Academy of Sciences of the United States of America. April. Vol. 110, No. 14, pp. 5695-5700.
[4] Jung, C. M. et al. 2011. Energy expenditure during sleep, sleep deprivation and sleep following sleep deprivation in adult humans. The Journal of Physiology. January. Vol. 589, Part 1, pp. 235-244.
[5] Koijma, M. et al. 1999. Ghrelin is a growth-hormone-releasing acylated peptide from stomach. Nature. December. Vol. 402, No. 6762, pp. 656-660.
[6] Pagel, J. F. et al. 2009. Excessive Daytime Sleepiness. American Family Physician. March. Vol. 79, No. 5, pp. 391-396.
[7] Patel, S. R. & Hu, F. B. 2008. Short sleep duration and weight gain: a systematic review. Obesity (Silver Spring). March. Vo. 16, No. 3, pp. 643-653.
[8] Schmid, S. M. et al. 2009. Short-term sleep loss decreases physical activity under free-living conditions but does not increase food intake under time-deprived laboratory conditions in healthy men. American Journal of Clinical Nutrition. December. Vol. 90, No. 6, pp. 1475-1482.
[9] Sivak, M. 2006. Sleeping more as a way to lose weight. Obesity Review. August. Vol. 7, No. 3, pp. 295-296.
[10] Spiegel et al. 2004. Brief communication: Sleep curtailment in healthy young men is associated with decreased leptin levels, elevated ghrelin levels, and increased hunger and appetite. Annals of Internal Medicine. December. Vol. 141, No. 11, pp. 846-850.
[11] Spiegel et al. 2004. Leptin Levels Are Dependent on Sleep Duration: Relationships with Sympathovagal Balance, Carbohydrate Regulation, Cortisol, and Thyrotropin. The Journal of Clinical Endocrinology. November. Vol. 89, No. 11, pp. 5762–577.
[12] St. Onge, M. P. 2017. Sleep–obesity relation: underlying mechanisms and consequences for treatment. Obesity Reviews. February. Supplement 1, pp. 34-39.
[13] Taheri et al. 2004. Short Sleep Duration Is Associated with Reduced Leptin, Elevated Ghrelin, and Increased Body Mass Index. PLoS Medicine. December. Vol. 1, No. 3, e62.
[14]Van Dongen, H. et al. 2003. The cumulative cost of additional wakefulness: dose-response effects on neurobehavioral functions and sleep physiology from chronic sleep restriction and total sleep deprivation. Sleep. Vol. 26, No. 2, pp.17–126.
[15] Weaver, T. E. et al. 1997. An instrument to measure functional status outcomes for disorders of excessive sleepiness. Sleep. October. Vol. 20, No. 10, pp. 835–843.
[16] Watson, N. F. et al. 2015. Recommended Amount of Sleep for a Healthy Adult: A Joint Consensus Statement of the American Academy of Sleep Medicine and Sleep Research Society. Sleep. Vo. 38, No. 6, pp. 843-844.
Did you find this content valuable?
Add yourself to our community to be notified of future content.





